link to F gas regulation: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=OJ:L_202400573
the following list has been shared:
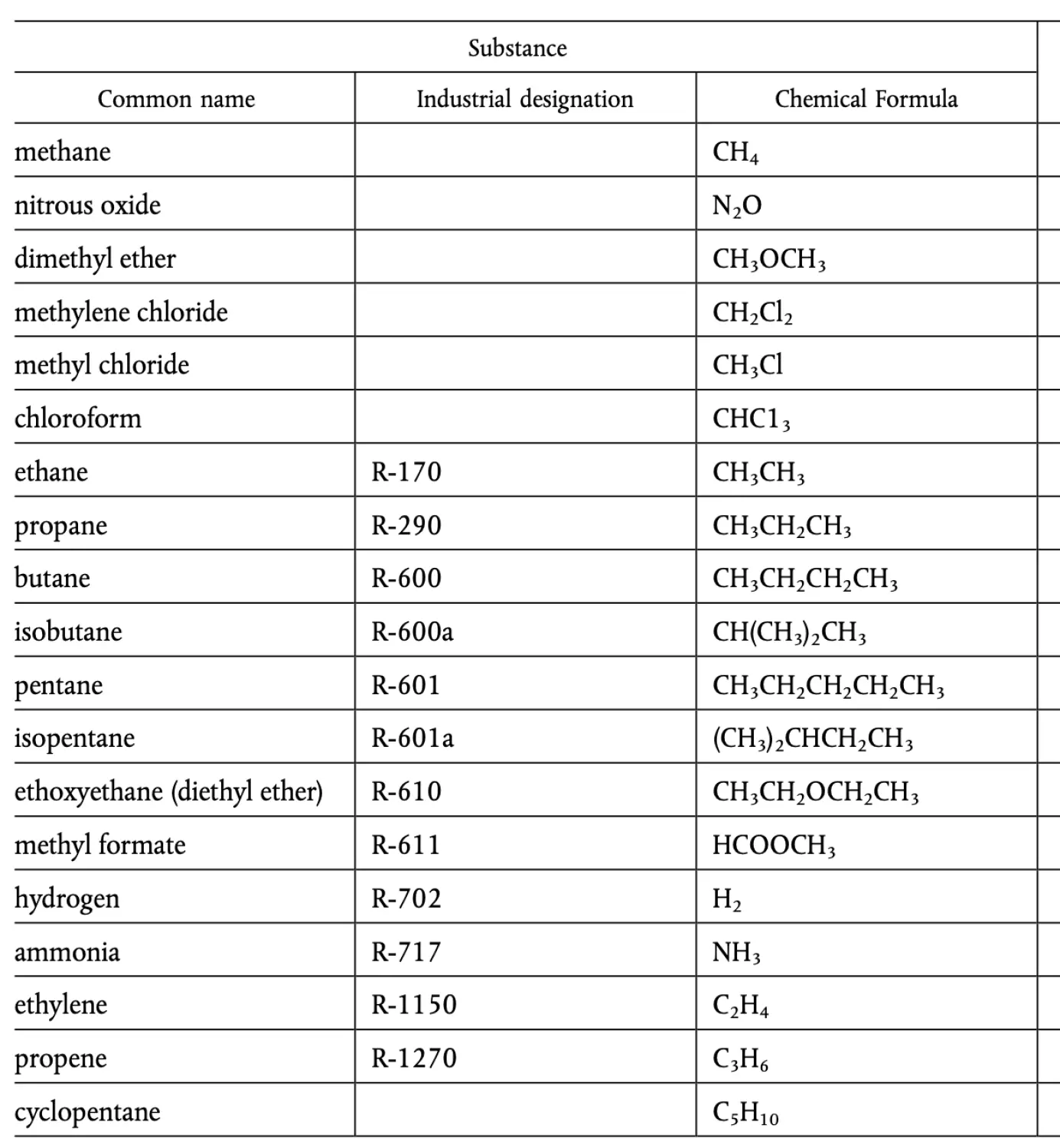
The following substances from the list have explosive properties or can form explosive mixtures:
- Methane (CH₄) – Although it is typically not classified as explosive by itself, methane can form explosive mixtures with air. It is highly flammable and explosive in confined spaces.
- Nitrous oxide (N₂O) – While not explosive in the traditional sense, nitrous oxide can support combustion and can be dangerous under certain conditions.
- Dimethyl ether (CH₃OCH₃) – It is flammable and can form explosive mixtures with air.
- Methylene chloride (CH₂Cl₂) – It is generally not classified as highly explosive, but it can be hazardous in certain conditions, especially under high pressure or when mixed with other substances.
- Methyl chloride (CH₃Cl) – It is flammable and can form explosive mixtures with air.
- Chloroform (CHCl₃) – While not typically explosive, chloroform can form dangerous compounds under specific conditions.
- Ethane (R-170) – It is flammable and can form explosive mixtures with air.
- Propane (R-290) – It is highly flammable and can form explosive mixtures with air.
- Butane (R-600) – It is flammable and can form explosive mixtures with air.
- Isobutane (R-600a) – It is flammable and can form explosive mixtures with air.
- Pentane (R-601) – It is highly flammable and can form explosive mixtures with air.
- Isopentane (R-601a) – It is flammable and can form explosive mixtures with air.
- Ethoxyethane (diethyl ether) (R-610) – Highly flammable and can form explosive mixtures with air. It is particularly hazardous due to its volatility.
- Methyl formate (R-611) – It is flammable and can form explosive mixtures with air.
- Hydrogen (R-702) – Hydrogen is highly explosive when mixed with air and in certain concentrations.
- Ammonia (R-717) – While ammonia is not highly explosive by itself, it can form explosive mixtures with air under certain conditions.
- Ethylene (R-1150) – It is flammable and can form explosive mixtures with air.
- Propene (R-1270) – It is flammable and can form explosive mixtures with air.
- Cyclopentane (C₅H₁₀) – It is highly flammable and can form explosive mixtures with air.
Notes:
- Explosive or flammable substances: Methane, Nitrous oxide, Dimethyl ether, Methyl chloride, Ethane, Propane, Butane, Isobutane, Pentane, Isopentane, Ethoxyethane, Methyl formate, Hydrogen, Ethylene, Propene, Cyclopentane.
- Not typically explosive: Methylene chloride, Chloroform, Ammonia(!).
- Note to Ammonia: it is flammable and can form explosive mixtures with air, making it subject to explosion protection (Ex) standards, especially in closed systems or areas with potential for leaks.
--- Here is a breakdown of whether the substances listed can be used as refrigerants:
Can be used for refrigeration:
- Methane (CH₄) – While methane is used in some specialized refrigeration systems (particularly in natural gas and low-temperature refrigeration), it's not commonly used due to its flammability.
- Dimethyl ether (CH₃OCH₃) – Can be used as a refrigerant due to its low boiling point and relatively low GWP. It is often considered as an alternative to some fluorinated refrigerants.
- Ethane (R-170) – Can be used in refrigeration, particularly in low-temperature applications, but is flammable.
- Propane (R-290) – Widely used as a natural refrigerant, especially in commercial refrigeration systems, because of its low GWP and efficiency. However, it is flammable.
- Butane (R-600) – Used in refrigeration, particularly in domestic refrigeration. It's flammable, which limits some of its applications.
- Isobutane (R-600a) – Commonly used in refrigeration (especially in small refrigeration units) due to its low environmental impact (low GWP), but it's flammable.
- Pentane (R-601) – Can be used as a refrigerant, particularly in industrial applications like refrigeration systems that use hydrocarbon-based refrigerants. It is also flammable.
- Isopentane (R-601a) – Similar to pentane and is used in refrigeration systems, especially where hydrocarbons are preferred due to their lower GWP.
- Ethoxyethane (diethyl ether, R-610) – Can be used as a refrigerant, though it is more commonly considered for specific low-temperature or experimental refrigeration applications.
- Methyl formate (R-611) – Can be used in refrigeration, especially in low-temperature systems, due to its low boiling point and relatively low GWP.
- Hydrogen (R-702) – Can be used as a refrigerant in some specialized high-efficiency systems, though its flammability and low boiling point make it less common.
- Ethylene (R-1150) – Used in industrial refrigeration, especially in low-temperature applications. It is flammable but has a low GWP.
- Propene (R-1270) – Used in some industrial refrigeration systems, with a focus on its low GWP. It's flammable.
- Cyclopentane (C₅H₁₀) – Used as a refrigerant, particularly in foam-blowing and refrigeration systems. It's flammable but has a low GWP.
Generally not used in refrigeration or less common:
- Nitrous oxide (N₂O) – While it has a high GWP, it is not typically used as a refrigerant due to its properties.
- Methylene chloride (CH₂Cl₂) – Not commonly used as a refrigerant; it is more often found in industrial cleaning or other chemical processes.
- Methyl chloride (CH₃Cl) – It can be used as a refrigerant in certain specific applications but is not as common.
- Chloroform (CHCl₃) – It is not used as a refrigerant due to its toxicity and environmental concerns.
Not suitable due to safety concerns:
- Ammonia (R-717) – While ammonia is widely used in large-scale industrial refrigeration systems, particularly for cooling, it is toxic and requires careful handling due to its hazardous properties. It is not commonly used in residential or small-scale systems.
Summary:
- Widely used refrigerants: Propane (R-290), Isobutane (R-600a), Ethane (R-170), Cyclopentane (C₅H₁₀).
- Can be used in specialized or low-temperature applications: Methane, Dimethyl ether, Pentane, Isopentane, Ethylene (R-1150), Methyl formate, Propene.
- Less commonly used, but possible: Hydrogen, Methylene chloride, Methyl chloride.
- Not typically used: Nitrous oxide, Chloroform, Ammonia (for smaller systems).
--- the below ones shall be with pre-cautions
- Propane (R-290) – Flammable and can form explosive mixtures with air. It is widely used as a refrigerant, but due to its flammability, it requires careful handling and proper ventilation to avoid explosions.
- Isobutane (R-600a) – Flammable and can form explosive mixtures with air. Like propane, it is commonly used as a refrigerant in small refrigeration systems, but its flammability is a major safety concern.
- Ethane (R-170) – Flammable and can form explosive mixtures with air. It is used in some refrigeration applications, but due to its flammability, it requires special precautions when used in confined spaces.
- Cyclopentane (C₅H₁₀) – Flammable and can form explosive mixtures with air. It is often used in foam-blowing and refrigeration applications but is also highly flammable, which can pose safety risks.
Note: GWP - global warming potential
Keep up the good work!
Arpad
veress@exprofessional.com


